|
 |
Turquoise |
 |
Did you know that
turquoise is
an opaque, blue-to-green
mineral
that is a
hydrous
phosphate
of
copper
and
aluminium,
with the
chemical formula
CuAl6(PO4)4(OH)8·4H2O.
It is rare and valuable in finer grades and has been prized as a
gem and
ornamental stone for thousands of years owing to its unique hue.
In recent times, turquoise, like most other opaque gems, has
been devalued by the introduction of treatments, imitations, and
synthetics onto the market.
I didn't either, and I also didn't know that the
Wikipedia
article on turquoise was written by someone who spells
"aluminum" "aluminium." I wonder if that's the subject of
a spelling war. Another thing I didn't know about
turquoise:
Being a
phosphate
mineral, turquoise is inherently fragile and sensitive to
solvents;
perfume
and other
cosmetics
will attack the finish and may alter the colour of turquoise
gems, as will skin oils, as will most commercial jewelry
cleaning fluids. Prolonged exposure to direct sunlight may also
discolour or dehydrate turquoise. Care should therefore be taken
when wearing such jewels: cosmetics, including
sunscreen
and
hair spray,
should be applied before putting on turquoise jewellery, and
they should not be worn to a beach or other sun-bathed
environment.
If I had known all that I just read in the Wikipedia
article before I conducted my experiment, I would have saved Trish
Garland some turquoise chips, the swimming pool at the gymnasium a PLUCO
full of water, and myself some time, effort, and worry. And there
might have been an entirely different blog for today, or, possibly none
at all.
What Experiment?
This experiment:
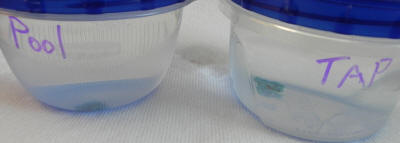
The result: It appears that there was no
noticeable difference between the control turquoise chip and its
companion experimental subject.
The Remaining Question
That would be: Why did I perform this experiment?
Although I have exhibited unabashed interest in the periodic table of
the elements, and have spotted a large number of
chemical names in the Wall Street
Journal, I haven't been concerned with gems or precious stones, and
certainly not obsessed to the extent of conducting experiments with
them. The answer is a simple one and is related to that most basic
of human aversions: pain.
"Getting with the program" in Sedona, I bought a jewelry
item of the "ring" persuasion. I've never worn any jewelry, ever,
and so this became a learning experience for me. As I would have
suspected had I ever spent a moment thinking about it, rings come in
"sizes," just like clothing. (Which I have and continue to wear,
although I give it about the same amount of thought as I did jewelry.)
What I did not and could not have realized before my trip to Garland's,
was this:
-
Rings of the correct size don't come off as easily
as they go on.
-
Rings with turquoise allegedly suffer damage and
discoloration in chlorinated water, to the extent that I was
cautioned to remove the ring every time I went swimming.
It took only minutes to convince me of the first.
Ow! Ow! Ow! Ow! I was not convinced of the
second, since I know people with turquoise jewelry who swim, and I
rarely hear a chorus of "Ow!"s emanating from the pool. So I made
a deal with the aforementioned Trish Garland: She would send me a
couple of chips of turquoise, and I would do the experiment detailed
above. Meanwhile, I had a 30-day return privilege on the ring if I
found it too inconvenient or downright painful to remove almost daily.
After performing my experiment and reading the Wikipedia
article, I'm guessing that the misconception about turquoise
discoloration is related to its sensitivity to sunscreen and sunlight—the
"beach environment" mentioned above. I decided to keep the ring,
which is really quite nice. And today I'm going to wear it in the
pool for the first time. Hopefully my experimental results will
translate to "real life."
|
NP:
"One Too Many Mornings"
The Association |
|
|
TotD
A bit out of sequence, but I'm allowed:
Terry's friend Juan maintains aquaria. Give him a call if
you're a 973 person with an aquarium and don't have a lot of
spare time or spare fish.
|
 |
|